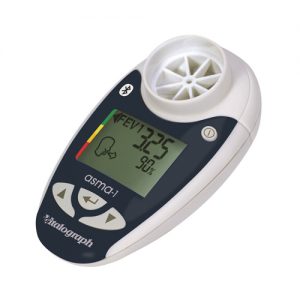
copd-6 bt
$405.00
For COPD case selection for spirometry with Bluetooth transmission of data
The copd-6™ bt combines all of the features of the copd-6 with the ability to transmit results via Bluetooth® using a paired device such as a mobile phone, PDA, PC or home hub. Session data and device ID may be transmitted to Vitalograph Reports or an API developers’ kit is available for providers of e-Diary, home hub and telemedicine solutions.
Features:
- Automatically sends data after each blow
- Can also send session data
- Time and date of each session
- Indicates blow quality and number of blows in each session
Transmits:
- FEV1, FEV6 and FEV1/FEV6
- Predicted values for the above
- Age, gender and height
- Device ID automatically transmitted with the data
- Reviews (83)
- Specifications
Be the first to review “copd-6 bt”
| Product: | Vitalograph copd-6 Bluetooth |
| Model Number | 4000 |
| Part Number | 40200, 40450, 40350, 40850 |
| Description | Vitalograph copd-6, Vitalograph copd-6 usb, Vitalograph copd-6 bluetooth, Vitalograph copd-6 serial |
| Parameters Displayed | FEV1/FEV6/ratio and % predicted |
| Quality of Blow Indicator | Yes |
| Obstructive Index | Mild, Moderate, Severe (FEV1% predicted) |
| COPD Classification | Normal (not COPD); Stage I, II, III & IV (FEV1% predicted) |
| Lung Age Indicator | Yes |
| Flow Impedance | Better than 0.15 kPa/L/s at 14 L/s |
| Operating Temperature Range | 17 – 37°C |
| Sensor | Stator rotor |
| Accuracy | Better than +/- 3% |
| Range | 0 – 9.99 L BTPS |
| Size | 113 x 63 x 48mm |
| Weight | 55g net |
| Display | Custom Liquid Crystal Display |
| Power | AAA batteries |
| Performance Standards | ISO 26782:2009 ISO 23747:2007 ATS/ERS 2005 |
| Safety Standards | IEC 60601-1:2005 |
| Medical Safety Standard | Medical Devices 93/42/EEC (as amended) |
| Designed and manufactured to following QA/GMP standards | ISO 13485:2003 FDA 21CFR820 |












Reviews
There are no reviews yet.