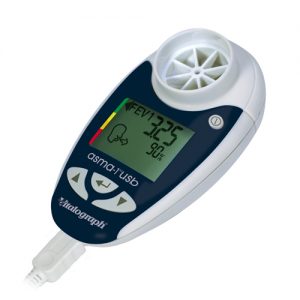
lung monitor bt
$450.00
For chronic disease management at home with Bluetooth transmission of data
The lung monitor bt combines all of the features of the lung monitor with the ability to transmit results via Bluetooth using a paired device such as a mobile phone, PDA, PC or home hub. Session data and device ID may be transmitted to Vitalograph Reports or an API developers’ kit is available for providers of e-Diary, home hub and telemedicine solutions.
This device can be used for respiratory monitoring in clinical trials, at home, at work, in primary or in secondary care.
- Automatically sends data after each blow
- Can also send memory dumps (up to 200 test sessions)
- Indicates blow quality and number of blows in each session
- FEV1, FEV6, FEF25-75 and FEV1/FEV6
- Personal best FEV1 as set in device
- Management zones as set in device
- Time and date of each session
- Device ID automatically transmitted with the data
- Reviews (83)
- Specification
Be the first to review “lung monitor bt”
| Product | Vitalograph lung monitor bluetooth |
| Model Number | 4000 |
| Part Number | 40500, 40400, 40750, 40950 |
| Description | Vitalograph lung monitor, Vitalograph lung monitor usb, Vitalograph lung monitor bluetooth, Vitalograph lung monitor serial |
| Parameters Displayed | FEV1, FEV6 , FEV1/FEV6 ratio and FEF |
| Memory | FEV1, FEV6 , FEV1/FEV6 ratio and FEF |
| Quality of Blow Indicator | Yes |
| Accuracy | Better than +/- 3% |
| Range | 0 – 9.99 L BTPS |
| Sensor | Stator rotor |
| Flow Impedance | Better than 0.15 kPa/L/s at 14 L/s |
| Power Supply | AAA batteries |
| Display | Custom liquid crystal display |
| Size | 113 x 63 x 48mm |
| Weight | 55g net |
| Performance standards | ISO 26782:2009 ISO 23747:2007 ATS/ERS Guidelines 2005 |
| Operating Temperature Range | 17 – 37°C |
| Management Zones | Factory set 3 zones Green/Yellow threshold 95% Yellow/Red threshold 90% Zones may be changed by the user And number of zones increased to 4 |
| Safety Standards | IEC 60601-1:2005 |
| Medical Safety Standard | Medical Devices Directive 93/42/EEC |
| Designed and manufactured to following QA/GMP standards | ISO 13485:2003 FDA 21CFR820 CMDR |














Reviews
There are no reviews yet.